
DUBAI: Commenting on the Group’s results, Roche CEO Severin Schwan said: “In the first half of the year, we achieved very strong results, driven by high demand for our new medicines. I am very pleased with the expedited approvals health authorities granted for Polivy and Rozlytrek. These medicines represent important treatment options for patients fighting cancer. Based on the performance in the first half of the year, we are increasing the outlook for the full-year 2019.” Sales are now expected to grow in the mid- to high-single digit range, at constant exchange rates. Core earnings per share are targeted to grow broadly in line with sales, at constant exchange rates.
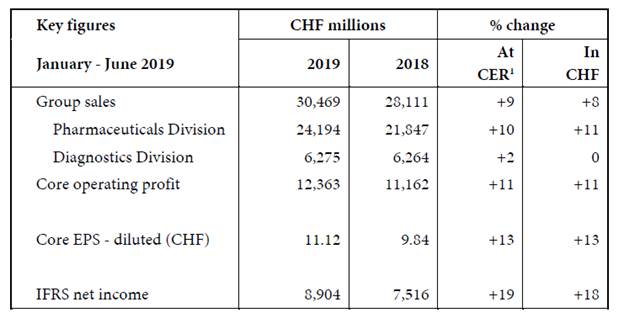
In the first half of 2019, Group sales rose 9% to CHF 30.5 billion and core EPS grew 13%, ahead of sales. Core operating profit increased 11%, reflecting the strong underlying business performance. IFRS net income increased 19%, due to the strong underlying core results and one-time effects resulting from a remeasurement of deferred tax positions as well as the release of acquisition related provisions.
Sales in the Pharmaceuticals Division increased 10% to CHF 24.2 billion. Key growth drivers were the multiple sclerosis medicine Ocrevus, the new haemophilia medicine Hemlibra and cancer medicines Tecentriq, Perjeta and Avastin. In the US, sales increased 14%, led by Ocrevus, Hemlibra, Tecentriq, Perjeta and Avastin. In Europe (-4%), sales were affected by the competition from biosimilars for Herceptin (-45%) and MabThera/Rituxan (-36%). In Japan, sales increased 9%, driven by recently launched products, including Hemlibra, Tecentriq and Perjeta. In the International region sales grew 17%, mainly driven by China with strong sales of Herceptin, Avastin and MabThera/Rituxan as well as launches of Alecensa and Perjeta.
Diagnostics Division sales increased 2% to CHF 6.3 billion. The business area Centralised and Point of Care Solutions (+3%) was the main contributor, led by the growth of the immunodiagnostics business. In regional terms, growth was reported in Asia-Pacific (+5%) and EMEA[1] (+3%). Sales declined in North America (-2%).
Core operating profit increased 11% in the Pharmaceuticals Division and 4% in the Diagnostics Division.
Important milestones for Roche medicines
The US Food and Drug Administration (FDA) granted accelerated approval for Polivy (polatuzumab vedotin-piiq) in combination with bendamustine plus Rituxan for the treatment of adults with relapsed or refractory diffuse large B-cell lymphoma who have received at least two prior therapies. The FDA’s Accelerated Approval Program allows the conditional approval of a medicine that fulfils an unmet medical need for a serious condition. In Japan, the Ministry of Health, Labour and Welfare (MHLW) approved Rozlytrek (entrectinib) for the treatment of adult and paediatric patients with neurotrophic tyrosine receptor kinase (NTRK) fusion-positive, advanced recurrent solid tumours. Rozlytrek is the first tumour-agnostic medicine to be approved in Japan that targets NTRK gene fusions, which have been identified in a range of hard-to-treat solid tumour types, including pancreatic, thyroid, salivary gland, breast, colorectal, and lung[2].
Roche Diagnostics – next generation solutions for individualised treatments
The Navify Tumor Board 2.0, the first collaboration product with GE Healthcare, was released for the markets. Incorporating medical image viewing and storage capabilities with other patient data, the product enables tumour boards – multi-disciplinary teams who determine treatment plans for cancer patients – to have a more comprehensive view of each patient in one place. The integration of GE Healthcare’s medical image viewer into Navify Tumor Board 2.0 enables radiologists to upload their patient records to the same dashboard where patient files from other disciplines in the cancer care team are stored. Having complete patient diagnostic information in one location helps specialists use the limited time they have during tumour boards to review all relevant files quickly and agree on the best possible treatment plan for each cancer patient.
Roche launched the Ventana HER2 Dual ISH DNA Probe Cocktail companion diagnostic test for breast and gastric cancer patients eligible for targeted therapy. HER2 – human epidermal growth factor receptor 2 – is an important biomarker in breast and gastric cancers, and its detection and inhibition can help manage these aggressive diseases more effectively. This test is designed to be completed within the same day, enabling clinicians to get results back quicker than with the most common methods of confirmatory testing for HER2. Results can be read using light transmission microscopy, eliminating the need for a specialised fluorescence microscope. The cobas MTB-RIF/INH test to detect resistance to antibiotics within tuberculosis DNA was launched in countries accepting the CE-mark. This assay is part of the mycobacteria test menu that includes the cobas MTB and cobas MAI tests for use on the cobas 6800/8800 Systems. This continues the expansion of the testing menu on the cobas 6800/8800 Systems, supporting true consolidation and efficient testing.
[1] EMEA = Europe, Middle East and Africa
[2] Demetri GD et al. Efficacy and Safety of Entrectinib in Patients with NTRK Fusion-Positive (NTRK-fp) Tumors: Pooled Analysis of Startrk-2, Startrk-1 and Alka-372-001. Presented at ESMO 2018; October 19-23, 2018; Munich, Germany. Abstract LBA17.











